1) Introduction
The cardiac outflow tract (OFT) is a transient structure at the arterial pole of the heart connecting the embryonic ventricles with the aortic sac. The OFT forms during heart looping from cardiac progenitor cells in pharyngeal mesoderm. At its maximal extension the OFT is a torsioned myocardial cylinder lined with endocardial cells, with orthogonal but contiguous proximal (conal) and distal (truncal) domains. The OFT is subsequently remodelled, concomitant with ventricular septation, during which process the OFT wall rotates and divides to generate the base of the ascending aorta and pulmonary trunk. OFT remodelling is associated with formation of an outlet septum, semilunar valve morphogenesis, coronary arteriogenesis and maturation of the myocardial and smooth muscle components of the base of the great arteries. Genetic or environmental perturbation of OFT elongation and remodelling lead to conotruncal congenital heart defects, accounting for 30% of all congenital heart defects (approximately 0.3% of live births) (Bruneau, 2008; Srivastava and Olson, 2000). Extensive anatomical, experimental and genetic studies of OFT morphogenesis have revealed the involvement of multiple cell types and signalling pathways in OFT development, suggesting that developmental complexity underlies the elevated incidence of OFT anomalies in man.
2) OFT formation
During heart tube elongation, myocardium is progressively added to the arterial pole of the heart from a progenitor cell population in pharyngeal mesoderm termed the second heart field (SHF; Figure 1A) (Buckingham et al., 2005; Kelly and Buckingham, 2002), (Cai et al., 2003). The SHF is positioned in mesoderm medial but contiguous to cells giving rise to the early heart tube; subsequently SHF cells underlie ventral foregut endoderm, constituting the dorsal wall of the pericardial cavity. The SHF contributes to the right ventricle in addition to the venous pole and AV regions of the heart; that population of progenitor cells contributing exclusively to the arterial pole is known as the anterior or secondary heart field. Critical transcriptional regulators of SHF development have been identified and include the transcription factors Isl1, Foxh1, Tbx1, Pitx2 and Mef2c (reviewed in (Buckingham et al., 2005)). Key properties of the SHF include its medial position, differentiation delay and elevated proliferation relative to cells that give rise to the early heart tube. These properties are regulated both intrinsically and by surrounding CNC cells and pharyngeal ectoderm and endoderm through the activity of signalling molecules of the fibroblast growth factor, bone morphogenetic protein, Hedgehog and Wnt pathways (Cohen et al., 2008; Goddeeris et al., 2007; Park et al., 2008; Prall et al., 2007). Together these signals define the niche maintaining SHF progenitor cells and regulating their progressive deployment during OFT extension (Rochais et al., 2009). The OFT is lined by endocardial cells also thought to derive from pharyngeal mesoderm (Laugwitz et al., 2008).
During heart tube elongation, myocardium is progressively added to the arterial pole of the heart from a progenitor cell population in pharyngeal mesoderm termed the second heart field (SHF; Figure 1A) (Buckingham et al., 2005; Kelly and Buckingham, 2002), (Cai et al., 2003). The SHF is positioned in mesoderm medial but contiguous to cells giving rise to the early heart tube; subsequently SHF cells underlie ventral foregut endoderm, constituting the dorsal wall of the pericardial cavity. The SHF contributes to the right ventricle in addition to the venous pole and AV regions of the heart; that population of progenitor cells contributing exclusively to the arterial pole is known as the anterior or secondary heart field. Critical transcriptional regulators of SHF development have been identified and include the transcription factors Isl1, Foxh1, Tbx1, Pitx2 and Mef2c (reviewed in (Buckingham et al., 2005)). Key properties of the SHF include its medial position, differentiation delay and elevated proliferation relative to cells that give rise to the early heart tube. These properties are regulated both intrinsically and by surrounding CNC cells and pharyngeal ectoderm and endoderm through the activity of signalling molecules of the fibroblast growth factor, bone morphogenetic protein, Hedgehog and Wnt pathways (Cohen et al., 2008; Goddeeris et al., 2007; Park et al., 2008; Prall et al., 2007). Together these signals define the niche maintaining SHF progenitor cells and regulating their progressive deployment during OFT extension (Rochais et al., 2009). The OFT is lined by endocardial cells also thought to derive from pharyngeal mesoderm (Laugwitz et al., 2008).
During OFT formation the arterial pole of the heart is displaced caudally in the pharyngeal region as pharyngeal arch morphogenesis and the formation of bilateral aortic arch arteries take place (Waldo et al., 2005b). CNC cells, derived from the dorsal neural tube, migrate through the caudal pharyngeal arches into the OFT of the heart during formation of the distal region of the OFT (Figure 1B) (Hutson and Kirby, 2003). In addition to a critical role in OFT remodelling, CNC cells play an early role in restricting proliferation and controlling deployment of the SHF (Waldo et al., 2005a). As CNC cells enter the distal OFT, endothelial cells in the proximal region of the OFT undergo a process of epithelial to mesenchymal transition in response to myocardial-derived signals, to form, together with invading neural crest cells, OFT cushions between the myocardial and endothelial layers (Sugishita et al., 2004b). Convergence of OFT cushions in a spiral structure separates laminar flow from the embryonic left and right ventricles. Cushion development depends on multiple intercellular signaling pathways including transforming growth factor b, fibroblast growth factor, bone morpogenetic protein, VEGF, Notch and non-canonical Wnt signaling (Wagner and Siddiqui, 2007). The OFT reaches its maximal length at midgestation in the mouse (E10.5) or the 4th week of development in man.
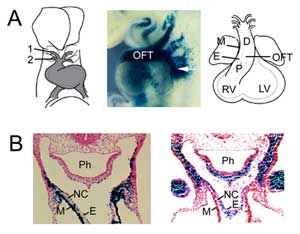
Figure 1
OFT formation. (A) The OFT at mouse embryonic day 8.5 is connected to the first (1) and second (2) aortic arch arteries (left) and extends by addition of second heart field progenitor cells from pharyngeal mesoderm (middle, arrow), visualised with an Fgf10 enhancer trap transgene at E9.5. The OFT attains maximal length at mouse embryonic day 10.5 and is comprised of proximal (P) and distal (D) regions. (B) Comparison of Fgf10 transgene expression in the OFT and second heart field (left) with neural crest derived cells (right) in the distal OFT at E9.5. Right hand panel reproduced from Jiang et al., 2001, Development 127:1607-16, with permission. M, myocardium; E, endocardium; RV, right ventricle; LV, left ventricle; Ph, pharynx; NC, neural crest derived cells.
3) OFT remodelling
During OFT remodelling the left ventricle acquires an independent outlet, the ascending aorta, providing a critical systemic conduit for oxygenated blood at birth. The cylindrical OFT is transformed into the base of the ascending aorta and pulmonary trunk (outlet of the right ventricle) concomitant with atrioventricular septation and aortic arch artery remodelling between days 10.5 and 14.5 of mouse gestation. OFT remodelling is a complex process with contributions from both genetics and haemodynamics (Yashiro et al., 2007). The critical roles of OFT cushion development and CNC cells in this process are established (Sugishita et al., 2004b). Proximal OFT myocardium is incorporated into the ventricular outlet and a process of invasive myocardialisation leads to muscularisation of the proximal outlet septum (Rana et al., 2007; van den Hoff et al., 2001). Ingrowth of the aorticopulmonary septum between the 4th and 6th aortic arch arteries separates the distal OFT into the ascending aorta and pulmonary trunk (Figure 2A). Smooth muscular walls develop at the base of the great arteries from the final contribution of the second heart field and more distally from CNCs (Sugishita et al., 2004b; Waldo et al., 2005b).
OFT septation is accompanied by rotation of the OFT wall in a counterclockwise direction resulting in alignment of the aorta with the left ventricle and pulmonary trunk with the right ventricle (Bajolle et al., 2006). This septation process is completed by convergence of the OFT, atrioventricular and ventricular septa (Figure 2A). The original distal OFT myocardial wall in part contributes to right ventricular subpulmonary myocardium and in part undergoes hypoxia-driven programmed cell death during ventriculoarterial alignment (Rana et al., 2007; Sugishita et al., 2004a). During the remodelling process, semilunar valves form at the angular junction of the proximal and distal regions of the OFT from the original and two intercalated OFT cushions (Anderson et al., 2003). Coronary arteriogenesis is the process by which the epicardially-derived coronary plexus selectively invades the base of the aorta above the two valve leaflets facing the pulmonary trunk, forming ostia connecting the coronary system with the ascending aorta (Figure 2B) (Tomanek, 2005).
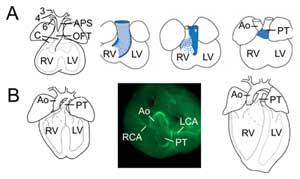
Figure 2.
Outflow tract remodelling. (A) At embryonic day 11.5 the OFT is connected to the third (3), fourth (4) and sixth (6) aortic arch arteries (left). The aorticopulmonary septum (APS) separates the distal OFT into the ascending aorta (Ao) and pulmonary trunk (PT) converging with OFT cushions (C). The right three images show progressive rotation of the myocardial wall during OFT septation: the dorsal wall at embryonic day 10.5 (blue) gives rise to ventral subpulmonary myocardium. (B) At embryonic day 14.5 OFT septation is complete and the aorta is connected to the left ventricle and pulmonary trunk to the right ventricle. In a superior view (middle) smooth muscle actin immunochemistry reveals the connections of the left (LCA) and right (RCA) coronary arteries to the base of the aorta. Structure of the definitive heart (right). RV, right ventricle; LV, left ventricle.
4) Conotruncal congenital heart defects
The underlying developmental complexity of OFT development is reflected in the high incidence of conotruncal anomalies in man (Bruneau, 2008; Srivastava and Olson, 2000). Failure of SHF or CNC deployment or of OFT remodelling contribute to the spectrum of conotruncal defects observed in human pathology and animal models (Figure 3) (Moon, 2008).
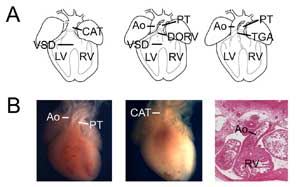
Figure 3.
Congenital heart defects. (A) Cartoons showing common arterial trunk (or persistant truncus arteriosus, CAT, left), double outlet right ventricle (DORV, middle) and transposition of the great arteries (TGA, right). VSD, ventricular septal defect; RV, right ventricle; LV, left ventricle, Ao, aorta; PT, pulmonary trunk. (B) Examples of mouse models of congenital heart defects. Control heart showing the aorta and pulmonary trunk (left) compared to a Tbx1 null heart (middle) with a single ventricular outlet (CAT). Example of a transposition-type double outlet right ventricle showing the aorta connected with the right ventricle in a Tbx3 null heart (right).
Whereas severe impairment of SHF development leads to failure of heart tube elongation, milder defects result in ventriculoarterial alignment anomalies and failure of the aorta to establish an independent connection with the left ventricle (Abu-Issa and Kirby, 2007). These defects may be intrinsic to the SHF or affect surrounding cell types (CNC or pharyngeal epithelia) that control the SHF progenitor cell niche. Such defects may result in double outlet right ventricle, overriding aorta, pulmonary hypoplasia, pulmonary atresia and tetralogy of Fallot. Loss or reduction of the CNC leads to failure or delay of OFT septation resulting in persistant truncus arteriosus or double outlet right ventricle; CNC deficiency also indirectly affects SHF development resulting in a composite alignment and septation phenotype (Hutson and Kirby, 2003). Failure of rotation of the OFT wall during OFT remodelling also results in alignment defects including transposition of the great arteries, double outlet right ventricle and overriding aorta, commonly subsequent to defects in embryonic laterality (Bajolle et al., 2006; Bamforth et al., 2001). Defects in formation of the OFT septum can also result from later failure of myocardial wall differentiation and anomalies in processes essential for remodelling such as programmed cell death or myocardialisation (Park et al., 2008). Intrinsic defects in OFT endocardial cells, as well as haemodynamic changes, have also been demonstrated to lead to OFT anomalies 16, (Zhang et al., 2009), (Bartman and Hove, 2005; Yashiro et al., 2007). Finally, abnormal proximal coronary artery patterning is frequently associated with conotruncal defects and, in isolation, is a significant cause of sudden cardiac death (Angelini et al., 2002). Among current research goals are a better understanding of the etiology of conotruncal defects through the identification of causative genetic mutations and modifiers by genome wide screens and dissection of the relative contribution of genetic and epigenetic players in OFT formation and remodelling using animal models. These studies will provide insights into the signaling pathways and cellular processes that drive OFT morphogenesis and are the targets of disease mechanisms.
Acknowledgements
Thank you to Profs. Diego Franco and Deborah Henderson for comments on the manuscript. RGK acknowledges support of the Inserm Avenir program, the Agence National de la Recherche, the CNRS and EU FP6 Heart Repair.

 Our mission: To reduce the burden of cardiovascular disease.
Our mission: To reduce the burden of cardiovascular disease.