1 - Definition
ARVD is a disease of the heart muscle associated with ventricular arrhythmias and sudden death. It is characterised by structural and functional abnormalities of the right ventricle caused by the replacement of the myocardium by fatty and fibrous tissue. The sites of involvement of anatomic abnormalities are found in the so-called triangle of dysplasia (the right ventricular subtricuspid areas, the apex and the infundibulum)(4).
Arrythmogenic right ventricular dysplasia (ARVD) is an inherited disease, typically inherited as an autosomal dominant trait with variable penetrance and incomplete expression(1). There is an autosomal recessive variant associated with palmoplantar keratosis and wally hair named Naxos disease.
2 - Prevalence
The prevalence in the general population is approximately from 1:2500 to 1:5000
Nevertheless, itis dependent on geographic circumstances, for example in certain regions of Italy (Padua, Venice) and Greece (island of Naxos), the prevalence is increased(2). It accounts for 5% to 10% of unexplained sudden cardiac deaths in individuals under65 years(3).
It occurs in young adults with a male to female ratio of 2,7/1. After hypertrophic heart disease, it is the number one cause of sudden cardiac death in young people.
3 - Diagnosis
Diagnosis of ARVD is often difficult because there is no single test. Diagnosis is based on the presence of structural, histological, electrocardiographic and genetic factors according to the 1994 Task Force Report by McKenna et al (5) (table 1) and a modification of the Task Force by Homid et al (6) (table 2) to increase the diagnostic sensitivity of first degree family members for early detection of the disease.
A) Clinical history
First of all, we’ll start with the easier tool, that is, clinical history.
Clinical presentation varies from asymptomatic forms to palpitations, fatigue, syncope or even cardiac arrest usually during exercise. These symptoms are due to ventricular ectopic beats, sustained ventricular tachycardia of left bundle branch block configuration or right ventricular failure(4).
We can identify four stages in the natural history of this disease(7):
a) the early or silent phase, it is a subclinical phase with concealed structural abnormalities
b) the unstable phase with electrical disorder
c) the right ventricular failure phase
d) the final phase with progressive biventricular failure, mimicking dilated cardiomyopathy
B) Diagnostic test
1.- Electrocardiogram
There are several ECG features in the criteria diagnosis of ARVD:
a) T wave inversions in V1 through V3 (minor diagnostic criterion, but one of most common ECG abnormality present in 85% of patients (8)
b) QRS duration = 110 ms in V1 through V3
c) Epsilon wave (electric potentials after the end of the QRS complex). It is a major diagnostic criterion found in up to 30% of cases of ARVD.
Other ECG markers of ARVD have been reported: QRS and QT dispersion, parietal block defined as a QRS duration in leads V1 through V3 that exceeds the QRS duration in lead V6 by > 25 ms, a prolonged S-wave upstroke in V1 through V3 = 55 ms (it was seen as the most prevalent ECG feature in 95% of ARVD8
2.- Myocardial imaging
2.1 Echocardiography
It is the non-invasive imaging technique most widely used but it is not always the optimal imaging technique in obese patients and in patients with pulmonary emphysema.
Echocardiography is the initial diagnostic approach in patients suspected of having ARVD. Principal findings are (9):
- right ventricular dilation and hypokinesia
- isolated dilatation of the right ventricular outflow tract
- increased reflectivity of the moderator band
- end-diastolic aneurysms
- akinesis-dyskinesis of the inferobasal segment and the right ventricular apex
- prominent apical trabeculae
2.2 Right ventricular contrast angiography
This technique is considered the reference standard for the diagnosis of ARVD (10). It consists of akinetic-dyskinetic areas localised in the anatomic triangle of dysplasia. However, due to an invasive technique, X-ray exposure, the interobserver variability, this method is not widely used (9).
2.3 Computed tomography
Computed tomography is capable of diagnosing patients with ARVD. Dery et al11, were the first to demonstrate a dilated hypokinetic right ventricle in a patient with ARVD.
Findings of ARVD on electron-beam computed tomography are7:
- the presence of epicardial fat or intramyocardial fat deposits
- conspicuous trabeculations with low attenuation
- dilated hypokinetic right ventricle
- scalloped appearance of the right ventricular wall
Currently, computed tomography is not the optional imaging modality for initial screening due to high radiation burden.
2.4 Cardiovascular magnetic resonance imaging
Magnetic resonance (MR) is an excellent tool for visualising the right ventricle, it allows a three-dimensional evaluation of ventricular anatomy, volumes and when compared against other techniques it can better recognise the replacement of myocardial fatty and fibrofatty tissue, although fatty infiltration of the right ventricle is not exclusive of ARVD, as it occurs in more than 50% of normal hearts in elderly people. However the presence of transmural fatty replacement or diffuse thinning of the right ventricle myocardium should be considered to be a major criterion for the diagnosis of ARVD.
MR can also be used to assess both systolic and diastolic function. Several studies have addressed the presence of right ventricular diastolic dysfunction as an early marker of the disease (12)
The typical criteria that can be demonstrated with MR are:
- presence of high-signal intensity areas indicating the substitution of myocardium by fat (major criterion)
- fibrofatty replacement which leads to diffuse thinning of the right ventricular myocardium (major criterion)
- aneurysm of the right ventricle and right ventricular outflow tract (major criterion)
- dilatation of the right ventricle and right ventricular outflow tract (when severe, major criterion; when mild, minor criterion)
- regional contraction abnormalities (minor criterion)
- global systolic dysfunction (major criterion) and global diastolic dysfunction (minor criterion)
Cardiovascular magnetic resonance imaging provides important anatomic, morphologic, functional and flow-dynamic criteria for diagnosing ARVD although the diagnosis of ARVD must be made based on Task Force criteria and not on structural abnormalities only.
2.5 Endomyocardial biopsy
Histological diagnosis is definitive, however endomyocardial biopsy is controversial because of the segmental nature of the disease and the samples are usually obtained from the septum (13). Complications can occur such as tamponade and perforation.
4 - Management of patients with ARVD
Before treatment, we have to know predictors of mortality and risk stratification. The study of Hulot et al (14) found that at least 1 episode of left bundle-branch block ventricular tachycardia, clinical signs of right ventricular failure and left ventricular dysfunction were associated with cardiovascular deaths.
The problem is that the appearance of sudden death is not related to disease progression and sudden death could be the first manifestation of the disease.
1.- Antiarrhythmic drugs
Antiarrhythmic drugs are the initial and most commonly used therapy. Beta-adrenergic blocking agents are recommended to reduce adrenergic stimulated arrhythmia.
The most successful drug is sotalol. Sotalol was more effective than beta-blocking agents or amiodarone in patients with inducible and non inducible ventricular tachycardia (VT) administered in dosages ranging from 320 to 480 mg/day (sotalol prevented VT during programmed ventricular stimulation in 68% whereas amiodarone 26% and class Ia and Ib 5,6% and class Ic only in 3% of the patients) (15).
2.- Catheter ablation
Radiofrequency ablation (RF) is used in cases of drug refractory/intolerance or incessant ventricular tachycardia. The goal of RF ablation is to eliminate conduction pathways. It is associated with complete success in only 30 to 65% of cases. Due to the progressive and diffuse nature of the disease, it isdifficult to abolish multiple arrhythmogenic foci16.
The patologic substrate is recorded during electrophysiologic study as a low amplitude, fractionated endocardial signal reflecting diminished local endocardial conduction velocity7.
3.- Implantable cardioverter-defibrillator therapy
Patients considered at high risk for sudden cardiac death should receive an implantable cardioverter-defibrillator (ICD). They are those who 1) have been resuscitated from cardiac arrest with a history of syncope, 2) have threatening arrhythmias that are not completely suppressed by antiarrhythmic drug therapy and 3) with a family history of cardiac arrest in first degree relatives (primary prevention).
ICDs work by providing antitachycardia pacing and defibrillation shocks when arrhythmias occur.
ICD therapy is feasible and safe in patients with ARVD with a low incidence of short term and long term complications. This tool has an important role in the treatment of ventricular arrhythmias, more than three quarters of ARVD patients received appropriate ICD therapy during an average of 3,5 years of follow up (17)
However, there can be complications of ICD therapy as result of the replacement of right ventricle myocardium with fat and fibrotic tissue. These include perforation caused by thinning of the right ventricle wall, difficulty in lead placement to inadequate R wave amplitudes or high pacing thresholds, inadequate sensing or pacing during follow-up resulting from disease progression and failure to terminate ventricular arrhythmias to rising defibrillation thresholds (18).
4.- Cardiac failure treatment
When right ventricular or biventricular failure appear, treatment consists of the current therapy for heart failure including diuretics, beta blocking agents, angiotensin-converting enzyme inhibitors and anticoagulants.
Curative therapy in case of refractory congestive heart failure and/or arrhythmias is cardiac transplantation.
Figure 1. Recording a post-excitation epsilon wave (arrows) in right precordial leads.
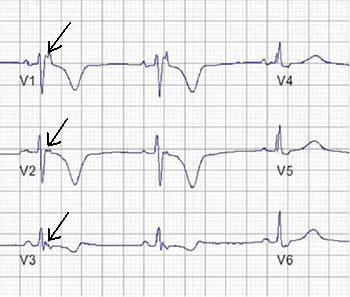
Figure 2. 12 lead ECG recording of VT with left bundle branch block morphology
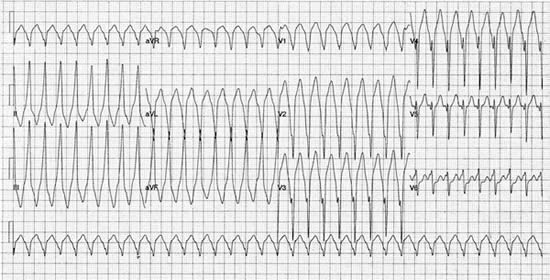
Table 1. Criteria for diagnosis of right ventricular dysplasiaI Global and/or regional dysfunction and structural alterations
| Major |
Severe dilatation and reduction of right ventricular ejection fraction with no (or only mild) LV impairment |
| Localised right ventricular aneurysms (akinetic or dyskinetic areas with diastolic bulging) | |
| Severe segmental dilatation of the right ventricle | |
| Minor | Mild global right ventricular dilatation and/or ejection fraction reduction with normal left ventricle |
| Mild segmental dilatation of the right ventricle | |
| Regional right ventricular hypokinesia |
II Tissue characterisation of walls
| Major | Fibrofatty replacement of myocardium on endomyocardial biopsy |
-
III Repolarisation abnormalities
| Minor | Inverted T waves in right precordial leads (V2 and V3) people aged more than 12 yr; in absence of right bundle branch block |
IV Depolarisation/conduction abnormalities
| Major | Epsilon waves or localised prolongation (>110 ms) of the QRS complex in right precordial leads (V1-V3) |
| Minor | Late potentials (signal averaged ECG) |
V Arrhythmias
| Minor | Left bundle branch block type ventricular tachycardia (sustained and non-sustained) (ECG, Holter, exercise testing). |
| Frequent ventricular extrasystoles (more than 1000/24h) |
VI Family history
| Major | Familial disease confirmed at necropsy or surgery |
| Minor | Familial history of premature sudden death (< 35 yr) due to suspected right ventricular dysplasia. |
| Familial history (clinical diagnosis based on present criteria) |
Table 2. Proposed modification of Task Force for the diagnosis of familial ARVD
ARVD in a first-degree relative plus one of the following:
| 1.- ECG | T-wave inversion in right precordial leads (V2 and V3) |
| 2.- SAECG | Late potentials seen on signal-average ECG (SAECG) |
| 3.- Arrhythmia | LBBB type VT on ECG, Holter monitoring or during exercise testing. Extrasystoles > 200 over a 24-h period |
| 4.- Structural or functional abnormality of RV | Mild global RV dilatation and/or EF reduction with normal LV Mild segmental dilatation of RV Regional RV hypokinesia |
The content of this article reflects the personal opinion of the author/s and is not necessarily the official position of the European Society of Cardiology



 Our mission: To reduce the burden of cardiovascular disease.
Our mission: To reduce the burden of cardiovascular disease.