Patients under chemotherapy treatment have an increased risk of developing cardiac toxicity. Until now, the main cardiac complications that have been described are left ventricular systolic dysfunction and heart failure. Continuous monitoring of a patient under this treatment is the best way to detect and establish an early diagnosis of cardiac toxicity. Echocardiography plays a definitive role for this assessment. (1)
Currently, the heart failure diagnosis of heart failure relies on clinical evaluation based on 1) history, 2) physical examination and 3) complementary tests. But heart failure symptoms are often non-specific. So, the use of adequate diagnostic methods to explore cardiac function is of critical importance in these patients.
Non-imaging diagnostic methods
The1) Eelectrocardiography should be routinely used to detect factors associated to with toxicity. Nevertheless, they these factors usually only appear only in advanced stages of the disease and their diagnostic accuracy is very limited. The
2) Eexercise stress testing is indicated in those patients with an intermediate pre-test probability of coronary artery disease and in those patients with doubts regarding a theirquestionable functional status. During and after treatment cessation of treatment, this test may be useful to accurately assess the functional status of a patient. The
3) Ccontinuous ambulatory electrocardiography is indicated when there is a need to clarify the diagnosis by detecting tachy or bradiarrythmias and in patients with sporadic symptoms to be related to arrhythmias such as syncope or palpitations. The Ttwenty-four-hours ambulatory blood pressure monitoring may be useful when considerable variability in the clinic measurement is found.
4) Electrophysiologic testing is recommended for in patients with previous myocardial infarction and symptoms suggestive of ventricular arrhythmias or left ventricular systolic dysfunction.
5) Tilt table testing is indicated in patients with unexplained syncope.
Echocardiography
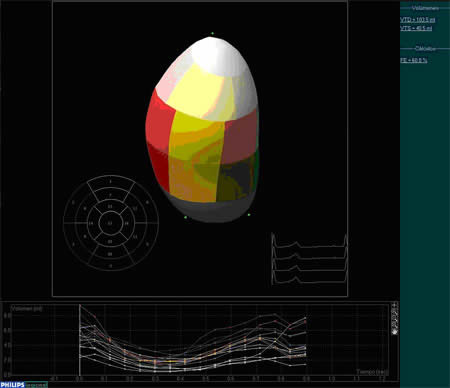
Echocardiography (Figure 1) is the cornerstone for of cardiac toxicity monitoring. Cardiac dysfunction is defined as a decrease in left ventricular ejection fraction of at least 10% to less than 55% or by a decrease of 5% in left ventricular ejection fraction in the presence of symptoms and signs of heart failure. Monitoring changes in left ventricular diastolic function could detect evidence of cardiotoxicity earlier than monitoring left ventricular ejection fraction. New methods based on Doppler-echocardiography such as tissue Doppler imaging and real-time 3D-echocardiography (Figure 2) are under evaluation. These techniques may enable more sensitive detection of early changes in myocardial function before those detected by traditional methods. (2,3,4)
Other diagnostic imaging methods (table 1)
Computed tomography-angiography may be useful in patients with intermediate probability of coronary artery disease specially if the electrocardiogram is not interpretable, if patient is unable to exercise or if the stress test provides equivocal results. The Ccardiac magnetic resonance may be used to evaluate left ventricular function prior to therapy and for monitoring the cardiac function during the cycles.
Serial multi-gated blood pool imaging (MUGA) is very reproducible but has the cost of radiation exposure to a young population. (5)
Pulmonary artery catheterisation, coronary angiography and angiography of peripheral blood vessels, should be recommended only in cases of discrepancies among the results of the non-invasive tests or when more precise information is needed in order to optimisze the management of patients. Until now, very few little information regarding the possible role of biochemical markers is available. (6,7)
Table 1: Cardiac imaging tool to assess left ventricular systolic function and their principal advantages and pitfalls.
| Pros | Cons | |
Echo-Doppler |
Cheap |
Variability |
| Available | Acoustic Window | |
| No Radiation | ||
MUGA |
Reproducibility |
Radiation |
| Ventriculography |
Invasive |
|
| CMR |
Accurate |
Expensive |
| Reproducible | Availibility | |
| No radiation | ||
| CT |
Accurate |
Expensive |
| Reproducible | Availibility | |
| Radiation |
CT: cardiac computed tomography; CMR: cardiac magnetic resonance; MUGA: multigated blood pool imaging.
Figure 1: Left ventricle M-mode echocardiography. Measurements of left ventricular end-diastolic diameter and end-systolic diameter are obtained and left ventricular ejection fraction is derived from them.
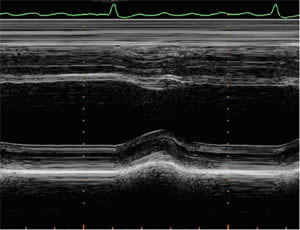
Figure 2: Real-time 3D echocardiography may play an interesting role in left ventricular function assessment. Their main advantages: global left ventricular assessment and improved accuracy.
The content of this article reflects the personal opinion of the author/s and is not necessarily the official position of the European Society of Cardiology




 Our mission: To reduce the burden of cardiovascular disease.
Our mission: To reduce the burden of cardiovascular disease.