Chagas disease: prevalence and transmission
According to the World Health Organisation, Chagas disease (American trypanosomiasis) represents the third largest tropical disease burden after 1) malaria: 40 million people infected in numerous tropical and subtropical countries and 2) schistosomiasis, also referred to as snail fever: 30 million infected in Asia, Africa, and South America. Approximately 10 million people are infected with Trypanosoma Cruzi, the etiological agent of Chagas disease. Cases are mostly in Latin America but others occur to other parts of the world as a result of migration, including in Europe, where expected prevalence of infected individuals is approximately 40,000.
Approximately 90% of transmission of Chagas disease occurs through an insect of certain species of triatoma within the reduviidae family, of which the triatoma infectans is the main vector (Fig. 1). These insects can carry the protozoan Trypanosoma cruzi, the parasite isolated in 1909 by the Brazilian physician Carlos Chagas and are almost exclusively established inside human dwellings and/or peridomestic structures. Insects are the intermediate hosts of the disease. Nocturnal, the bugs live in cracks and crevices of rural clay or mud houses and take blood meals on humans and animals while they are sleeping. Also referred to as "kissing bugs" they bite usually near the mouth or eyes, and defecate after feeding, dropping infective trypomastigote forms, which penetrate through the hole of the bite or solution of continuity left by scratching, or even by active penetration of the mouth and mucous and eyes. The bugs are responsible for transmitting disease in 90% of cases. They get the disease from biting animals or humans, which are both the primary hosts in which the parasite reaches maturity. Trypanosoma cruzi have been found in 150 species of animals that are an immense reservoir for the disease and 10 million infected humans. Death resulting from Chagas disease occurred in an estimated 10,000 people in the year 2008. (1)
Infection can also occur through blood transfusions, from mother to child via placenta, from ingestion of contaminated food and juices or from organ transplants. Globalisation has lead to increased migration of infected individuals to cities both within and outside Latin America, thus to non-endemic countries, mainly the United States, Alaska, Spain, Canada, Japan, Australia, France, Italy and the United Kingdom (2).
Manifestation and treatment
The acute phase of the disease lasts 4-8 weeks and is usually asymptomatic although a distinctive swelling of the eye, referred to as Ramona's sign that may develop at the beginning of the 1-2 week incubation period in the first days after the bite.
Death, from myocarditis and/or meningoencephalitis occurs in less than 5% to 10% of symptomatic patients. The acute phase may also occur due to reactivation of disease in patients with immunosuppressive states and in patients undergoing organ transplantation. During the acute or reactivation phase, diagnosis can be made with a search the protozoan or to study the host immune response by serum antibodies.
Standard treatment during the acute phase is benznidazole for a 60-90 day course, which tends to be better tolerated then nifurtimox. Nevertheless, both medications are expensive, have high risks of side effects, and cannot be used in pregnant women or those with renal or hepatic insufficiency. These anti-parasitic medications, if given early, can reduce the duration and severity of infection, but are only half effective in killing the organism itself. There is currently no approved treatment for chronic Chagas disease and there are currently no vaccines in use.
Sixty to 70% of those infected will never develop the disease and most often do not know they have been infected. They are said to have the indeterminate form. However 30% of all those infected, between 10 to 30 years after an initial infection that was left untreated or was unresponsive to parasitic elimination, will develop a chronic form which attacking internal organs will take on, in 20% of cases, a cardiac form, with or without ventricular dysfunction and in 10% to 15% of cases, a digestive or cardiodigestive form (3,4). During this stage, diagnosis is made after a study of the patient's clinical findings, and likelihood of being infected, such as having lived or visited an endemic country. Diagnosis is can be made by testing with at least two different serologic tests.
Typical manifestation of Chagas heart disease is dilated cardiomyopathy, congestive heart failure, arrhythmias, cardioembolism, and syncope. Here will focus on syncope.
Focus on syncope
Main causes for syncope in Chagasic disease are 1) neurological mechanism, 2) sinus node dysfunction, 3) atrioventricular block and 4) ventricular tachycardia.
- Neurally-mediated syncope can occur due to vasovagal reaction or orthostatic (or postural) hypotension, as in other Chagas-free individuals. Impairment of the autonomic nervous system, which precedes ventricular dysfunction, creates a situation of dysautonomia (4,6), and may be implicated in its pathophysiology.
*Diagnosis and treatment follow guidelines for syncope. (5). - Bradyarrhythmias are frequent causes and are due to sinus node dysfunction and atrioventricular (AV) conduction disturbances. A mononuclear cell infiltrate with edema, atrophy and necrosis of specialised fibers and replacement fibrosis, resulting in impairment of sinus node and AV node is described (4,7). Sinus node dysfunction is manifested by persistent sinus bradycardia or inadequate chronotropic response to exercise, sinoatrial block and sinus pause, and less frequently, bradycardia-tachycardia syndrome, probably due to autonomic denervation. It may also be secondary to antiarrhythmic drugs used to treat ventricular arrhythmias. The incidence of sinus node dysfunction observed by prolongation of the sinus node recovery varies from 3.8% to 18.1% (7-9). Another cause is AV conduction disorders (Fig.2), mostly in the infra-His location and frequently associated with right bundle branch block (7). These AV blocks were detected in 21% to 23% of chagasic patients with heart disease who underwent an electrophysiologic study (9,10). They can be intermittent and may occur with the progression of infranodal conduction system disease manifest by bifascicular block.
*Guidelines for pacemaker implantation in the context of bradyarrhythmias are to be followed (4). - Lastly, sustained ventricular tachycardia (SVT) can cause syncope in cardiac Chagas disease. The circuit of arrhythmia uses the areas of myocardial tissue fibrosis or aneurysms, the majority of the left ventricle, predominantly in the inferolateral region, followed by the septum and the apical and the mechanism is reentrant (11-15). Thus, although apical aneurysm is highly prevalent in this cardiomyopathy, a large proportion of ventricular tachycardia, approximately 80%, originates from the inferolateral region of the left ventricle, and only 8% originate in the apical region (13-16), perhaps due to the different mechanisms of reentry in the region inferolateral, with circuits in the endocardium, the mitral annulus and the epicardium. The most common morphology of SVT is right bundle branch block (Fig. 3). Another site of origin of SVT is the right ventricle, which may be responsible for 12% of ventricular tachycardia by endocardial mapping (16).
*The main objective of treatment of SVT is to improve quality of life by controlling symptoms and preventing sudden death. Treatment modalities include the use of antiarrhythmic drugs (mostly amiodarone), surgical ablation in selected patients, catheter ablation and implantable cardioverter-defibrillator (4) (Fig. 4).
Indications for prognosis:
- Unexplained syncope: Chagasic patients with unexplained syncope after a conventional evaluation in which ventricular tachycardia is induced by programmed ventricular stimulation, have a
* worse outcome, with a higher incidence of death and recurrence of syncope (17). - Patients with and without syncope and SVT: comparing chagasic patients with and without syncope and SVT, has shown that despite the higher syncope recurrence in the group with this initial presentation:
* the incidence of cardiac death was similar between the two groups (18). - Syncope and nonsustained ventricular tachycardia (NSVT) among cardiomyopathy chagasic patients with syncope and nonsustained ventricular tachycardia,
*syncope was not predictive of cardiac death during an average follow up of nearly 5 years (9). - Cardiomyopathy chagasic patients with NSVT and SVT: cardiomyopathy chagasic patients with NSVT and SVT showed
*a higher frequency of syncope in patients with SVT, but no association with mortality.
Figure 1: A haematophagous insect, which can become a vector for Trypanosoma cruzi by biting an infected animal or person. The bugs usually live in palm trees and rock crevices, but some live in human dwellings, typically hiding during the day in the cracked walls of mud or mud-brick houses, and coming out at night to feed.

Figure 2: Twelve-lead electrocardiogram of a 69-year-old male patient with chronic Chagas disease, (before pacemaker implantation) with manifestations of syncope without prodrome, in orthostatism and no trauma, showing low atrial rhythm and AV block 2:1 and ventricular rate of 37 beats per minute. In CM5 lead, there is a period of second-degree atrioventricular block Mobitz I. Patient underwent implantation of dual chamber pacemaker, with remission of symptoms.
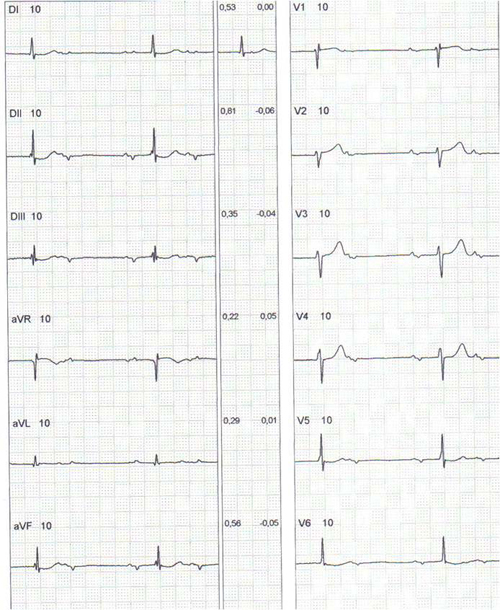
Figure 2b: A tracing with a long derivation, (before pacemaker application).
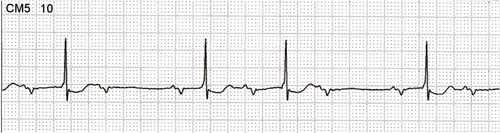
Figure 3: 12-lead electrocardiogram of 49-year-old male patient, with chronic Chagas disease, with symptoms of sudden palpitations and syncope, demonstrating sustained ventricular tachycardia with morphology of right bundle branch block, with periods of atrioventricular dissociation and ventricular rate of 230 beats per minute. The patient underwent successful defibrillation. By the echocardiogram, his ejection fraction of left ventricle was 0.47. He underwent implantation of a cardioverter-defibrillator.
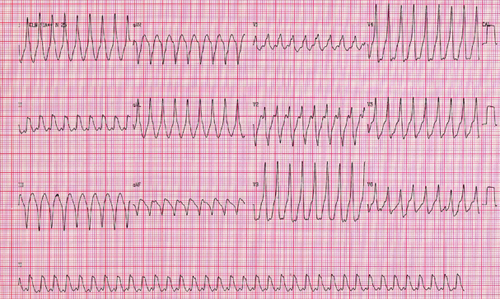
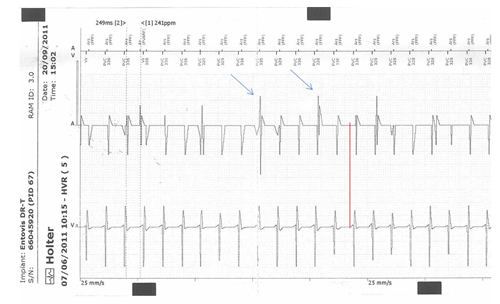
Figure 4: Intra-cardiac tracing recorded via the telemetry channels of a 56-year-old-female chronic Chagas disease patient. She underwent pacemaker implantation (DDDR mode) 8 years ago, due to advanced atrioventricular block, and generator change 4 months ago. She presented palpitations and presyncope with a duration up to 15 minutes, demonstrating telemetry ventricular tachycardia with a cycle frequency of 249 ms (241 beats per minute). There is ventriculoatrial conduction. The arrows show the pacemaker spikes. Patient was treated with furosemide, spironolactone, captopril, carvedilol, digoxin and amiodarone. By the echocardiogram, the ejection fraction was 0.27 without ventricular dyssynchrony, and the patient underwent implantation of a cardioverter-defibrillator, with control of symptoms.
Conclusion:
We have focused on sycope to show that sycopes' prognosis depends on its cause. Investigation and risk stratification allow us to establish appropriate treatment for symptom control and prevention of sudden cardiac death. Prognosis of syncope in patients with Chagas heart disease is not a predictor of cardiac death, and is relevant to a joint assessment with other variables (3,19,20).
In terms of a perspectives on the epidemic, large-scale vector control programs have been put in place since the 90's especially- spraying houses and roofs with insecticides to kill the insects, housing programs, and controls of blood and derivatives transfusion have been successful in containing transmission of Chagas disease in many countries of Latin America. However, the amazon deforestation causes the insects to fly to urban dwellings. Furthermore, a vast animal and human reservoir for the disease, with most those infected are unaware of their infection remain potential sources of transmission. Eradication without a vaccine seems difficult. In the meantime, globalisation of the disease seems inevitable (21).


 Our mission: To reduce the burden of cardiovascular disease.
Our mission: To reduce the burden of cardiovascular disease.